Tea is one of the most widely consumed beverages worldwide, particularly in China, where tea drinking can be traced back at least 1,500 years. Today, tea is the second most widely consumed drink in the world, after water.
The beverage is produced from the leaves of the shrub Camellia sinensis, which grows indigenously in China, Japan, India, and Thailand. There are two major varieties of the tea plant, Camellia sinensis var. sinensis, known as Chinese tea, and Camellia sinensis var. assamica, which is Assam, or Indian tea.
Over 3 million tons of tea are grown annually, 30 percent of which is produced in India, the world’s largest tea-producing country. Tea consumption is associated with a stimulant effect, due to the presence of caffeine, and a range of health benefits, partially attributed to the antioxidant activity of polyphenols contained in tea. Tea is also the focus of several traditional social rituals, such as the Japanese tea ceremony and British teatime, which contribute to its popularity.
Harvesting and Processing Tea Leaves
Tea is harvested by plucking the bud and the first two leaves of the Camellia sinensis plant. Depending on the methods used to process tea leaves, three different types of tea can then be produced: green, oolong, and black tea.
Generally, after harvesting, the leaves are rolled, disrupting the cellular compartmentation and bringing phenolic compounds into contact with the enzyme polyphenol oxidase. In the production of green tea, the rolled leaves are steamed or dried immediately to inactivate the enzyme and minimize oxidation. In contrast, to produce black tea, the rolled leaves undergo oxidation (fermentation) before drying. Oolong tea is produced similarly to black tea, deploying a shorter fermentation period.
Tea is known to have a highly complex chemical composition comprising diverse polyphenols, purine alkaloids, polysaccharides, amino acids, vitamins, lipids, and volatiles. The predominant polyphenols contained in green tea are catechins (flavan-3-ols) such as gallocatechin, epicatechin, epigallocatechin, epicatechin gallate, and epigallocatechin gallate. Epigallocatechin gallate is the most abundant catechin present in green tea. In the production of black tea, the monomeric catechins undergo oxidative polymerization to form the condensation products theaflavins and their polymers thearubigins.
Due to the complex composition of tea and the structural similarity of green tea phenolics, complete separation of the phenolic compounds in tea cannot be achieved using conventional one-dimensional liquid chromatography (1D-LC). However, separation power can be greatly increased using comprehensive two-dimensional liquid chromatography (2D-LC). 2D-LC is a technique in which two independent LC separations are applied to the sample. In comprehensive 2D-LC, the complete effluent following a first LC separation is injected onto a second column for further separation. This second dimension greatly increases the peak capacity and, as a result, the resolving power without increasing the analysis time.
This technical article demonstrates the comprehensive 2D-LC analysis of green and black tea. The quantification of the purine alkaloids, caffeine, and theobromine as well as the tea catechins enables a comparison of the composition of green and black tea.
Experimental Methods

Figure 1. Comprehensive 2D-LC separation of purine alkaloids and catechins (10 µg/mL each).
Image Credit: Agilent Technologies
Ten different samples of green tea and black tea were purchased from a German retail market. For this analysis, approximately 2 grams of the finely ground tea were extracted three times in acetone and water. The resulting extracts were each centrifuged to remove any particulate, and the resulting supernatants combined. A 100 microliter sample of this extract was then evaporated using a SpeedVac to dryness, and the resulting residue redissolved in 1 milliliter (mL) acetonitrile/water/acetic acid. The sample was then filtered before LC analysis.
LC analyses were performed using an Agilent 1290 Infinity II 2D-LC solution, deploying reversed-phase LC in both the first and second dimension. For this separation, an Agilent ZORBAX Eclipse plus C18 LC column was used for the first dimension and an Agilent Poroshell 120 Bonus-RP for the second dimension. Detection was achieved using an Agilent 1290 Infinity II diode array detector.
Method Validation

Table 1. Coefficients of linearity (R²) for calibration in the range from 2 to 100 µg/mL.
Image Credit: Agilent Technologies
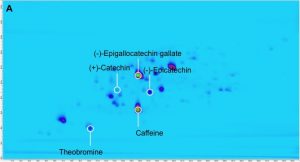
Figure 1 shows the separation of a 10 micrograms (μg) per mL mixture of the purine alkaloids, caffeine, theobromine, and theophylline as well as the catechins, catechin, epicatechin, and epigallocatechin gallate. For this complex mixture, 2D-LC setup enables a complete separation of the purine alkaloids and catechins. In the first-dimension separation, a coelution of caffeine and epigallocatechin gallate is observed, which is resolved in the second-dimension separation. Deploying only the second-dimension separation, catechin and epicatechin would coelute.
The precision of retention times and peak volumes were determined following multiple injections of this 10 μg per mL mixture of purine alkaloids and catechins (n=10). For these compounds, the second-dimension retention time precision was always below 2.5 percent and the peak volume precision was below 1 percent. These results demonstrated that the resolution and precision of 2D-LC is sufficient for the analysis of complex chemical compositions.
To quantify the levels of purine alkaloids and catechins in green and black tea, calibration curves were produced from each of the standard stock solutions. The response of caffeine, theobromine and theophylline, catechin, epicatechin, and epigallocatechin gallate at concentrations ranging from 2 to 100 µg per mL was measured. The coefficients of linearity produced for the purine alkaloids and catechins are shown in Table 1, confirming the excellent linearity achieved.
Analysis of Green and Black Tea Samples
Example chromatograms produced from a 2D-LC separation of one black tea sample and one green tea sample are shown in Figures 2a and 2b. Theophylline could not be detected in any of the analyzed tea samples. In green tea, the epigallocatechin gallate peak shows at a much higher intensity compared to black tea. In black tea, there are several peaks detected at retention times between 30 to 32 minutes in the first-dimension that are not visible in green tea. These peaks could originate from theaflavins and thearubigins present in black tea.
Ten different samples of green and black tea were analyzed, and Graph 1 shows the quantification results for purine alkaloids and catechins. As expected, the green tea samples generally contain higher amounts of the catechins epigallocatechin gallate and epicatechin than the black tea samples. This difference is due to the longer fermentation period used in the production of black teas. In which the monomeric catechins undergo oxidation polymerization to form theaflavins and their polymers thearubigins.

Graph 1. Quantification of purine alkaloids and catechins in green and black tea.
Image Credit: Agilent Technologies
Summary
This experiment demonstrates that a range of key phenolic compounds in complex tea samples can be separated using the resolving power of 2D-LC. This allowed precise assessment of the degree to which the catechins, epigallocatechin gallate, and epicatechin dominate in green tea samples in comparison to black tea samples. In comparison to 1D-LC, this 2D-LC approach revealed more information about the nature of black tea through the resolution of several peaks not significant in green tea.
Lee is global food market manager at Agilent Technologies, Inc. Reach him at [email protected].
ACCESS THE FULL VERSION OF THIS ARTICLE
To view this article and gain unlimited access to premium content on the FQ&S website, register for your FREE account. Build your profile and create a personalized experience today! Sign up is easy!
GET STARTED
Already have an account? LOGIN